
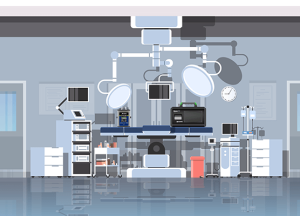
Translating Smoke Studies Into Fixed Probe Locations On Aseptic Filling Lines
Smoke studies are one of the best tools you have for deciding where your particle probes

Smoke studies are one of the best tools you have for deciding where your particle probes

In controlled environments like cleanrooms, laboratories, and manufacturing facilities, maintaining air quality and environmental conditions isn’t

In complex industries—from pharmaceutical cleanrooms to semiconductor facilities—monitoring systems are mission-critical. Yet, time and again we

Monitoring particle levels is crucial to maintaining cleanroom integrity and ensuring product quality across many industries.
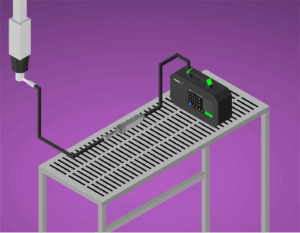
Keeping compressed gases clean and contaminant-free is a critical step in maintaining product quality and compliance,

At Lighthouse Worldwide Solutions®, we don’t just build products: we build partnerships with the people who

In highly regulated industries such as pharmaceuticals, biotechnology, and advanced manufacturing, contamination control is non-negotiable. Airborne

Remote particle counters are fixed units installed right inside your cleanroom at critical locations. Unlike handheld
After more than two decades, the world of cleanrooms just received a major refresh with the

King Mongkut’s University of Technology North Bangkok (KMUTNB) has always stood at the forefront of Thailand’s

When it comes to maintaining the integrity of cleanroom environments, human contamination remains the single greatest threat.

When it comes to viable sampling in cleanrooms and critical spaces, you’ve got two main media

The ActiveCount Remote Impactor Kit is designed to provide a flexible, modular solution for viable sampling

Ensuring Cleanliness and Product Quality Keeping precision parts free from contamination is crucial in industries like

In today’s world, precision and cleanliness are critical in industries like pharmaceuticals and biotechnology. Among the

Liquid particle counters are like silent guardians in industries where fluid purity is critical. From pharmaceuticals

Cleanrooms are essential controlled environments used in industries such as pharmaceuticals, biotechnology, and electronics manufacturing. Their

Particle monitoring is like having a super-sensitive detective on your team, keeping an eye on invisible

At Lighthouse Worldwide Solutions, we’re excited to introduce our latest innovation: the Active Count Remote Impactor

In 2024 our knowledge center webinar series provided a comprehensive exploration into the latest advancements and

Ultra Pure Water (UPW) is critical in industries where even the smallest impurities can have significant

At Lighthouse Worldwide Solutions, we’re excited to introduce our latest innovation: the ActiveCount Remote Impactor Kit.

Liquid particle counting is essential for maintaining public health and safety by monitoring the purity and

It’s actually not as complicated as it might sound at first. Think of GAMP as your

Airborne Particle Counters (APCs) play a critical role in monitoring and maintaining the cleanliness of environments

In a cleanroom, even the smallest particle can pose significant risks. Particle deposition occurs when airborne

Ensuring that the equipment used within cleanrooms is easy to wipe down and free from particle

Gas Sampling in Cleanrooms In cleanrooms, especially those classified ISO 5 or cleaner, maintaining contamination control

Overview Cleanrooms are essential in the pharmaceutical industry, providing controlled environments for the aseptic production of
Understanding High Pressure Diffusers High Pressure Diffusers (HPDs) are essential for maintaining the precision of particle

Understanding High Pressure Diffusers High Pressure Diffusers (HPDs) play a crucial role in ensuring the accuracy

As our world steadily moves towards a future dominated by electric vehicles (EVs), the safe and

In the realm of cleanroom operations, humans are both a crucial asset and a potential source

In the meticulous world of cleanroom operations, efficiency isn’t just a goal—it’s a necessity. From real-time

In the realm of sterile manufacturing processes, the Environmental Monitoring System (EMS) serves as a crucial

In the world of pharmaceutical manufacturing, biotechnology, semiconductor fabrication, and cleanroom environments, precision monitoring is critical
In the rapidly evolving landscape of pharmaceutical manufacturing, the integration of cutting-edge technologies is not just

Introducing a groundbreaking revolution in the world of liquid particle counting, we are thrilled to announce
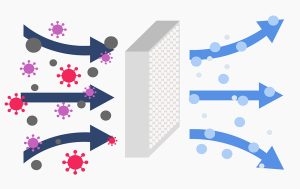
In the world of cleanrooms, where the highest standards of cleanliness and contamination control are paramount,

The rapid rise of electric vehicles (EVs) has brought about a surge in demand for high-quality
The 2022 update of the EU GMP Annex 1 has placed significant emphasis on the concept

Air Sampler impaction technology has been around for decades. However, the d50 is a little known

Introduction In the realm of pharmaceuticals, the pursuit of excellence in product quality is a never-ending

Introduction Cleanrooms in the Pharmaceutical industry are sanctuaries of precision, where the highest standards of cleanliness

Introduction In the high-stakes realm of Pharmaceutical manufacturing, precision and adherence to stringent cleanliness standards are

Introduction Airborne particle counters play a crucial role in ensuring the environmental integrity of pharmaceutical, bio-pharmaceutical, and
Semiconductor manufacturing is a highly intricate process, comprising numerous steps and stages, with each one presenting

Compliance with USP 788 standards doesn’t have to be a complex and laborious process. Recent advancements in liquid

The Vertex50: A Brief Introduction The Vertex50 is a liquid particle counter equipped with multiple size

Water quality plays a critical role in the pharmaceutical industry, where stringent requirements must be met

Pharmaceutical injectable aseptic injections are essential medical products that are administered through injections, directly into the body. The

In the meticulous realm of pharmaceuticals and controlled environments, precision is paramount. Particle counters emerge as

In the complex landscape of pharmaceutical manufacturing, where the safety and efficacy of medicinal products are

In our rapidly evolving world, environmental monitoring has become a critical aspect of safeguarding our planet’s

In 2022, GMP Annex 1 was revised to reflect the changing landscape of technology, discoveries, best

“Risk mitigation” is one of our favorite phrases. The best way to prevent yield loss, ensure

Environmental Monitoring Systems (EMSs) are important and hefty investments that require typically large amounts of infrastructure;

Aseptic manufacturing is one of the most difficult forms of manufacturing. From the processes to the
Semiconductors are the backbone of our world and the technology that runs our lives, businesses, health
Choosing a particle counter for semiconductor manufacturing is not a small decision. It is a massive
One of the most important things you can do during semiconductor manufacturing is practice proper risk

Quality control in semiconductor manufacturing is superficially simple: create consistent quality products. That statement is easy

Lighthouse Worldwide Solutions, a renowned global leader in the manufacturing of cutting-edge contamination control equipment, is

We talk a lot about life cycles, but have you used this approach to implementing a

GAMP5: another day, another acronym. So let’s break it down! GAMP5 stands for the Good Automated

Good Automated Manufacturing Practices (GAMP) has been defining IT validation since 1991. Since then, it has

GAMP5 refers to the fifth publication of Good Automated Manufacturing Practices published by the International Society

At this point, we are no stranger to global health crises. But these crises extend outside

In the wake of Pharma 4.0 – the term coined by ISPE to identify the fourth

The United States Pharmacopeia – National Formulary (USP-NF) is comprised of 6,800 monographs for over-the-counter and

The United States Pharmacopeia – National Formulary (USP-NF) serves as a resource for drug manufacturers, researchers,
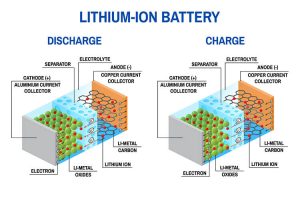
The article is mainly focused on the impact, causes and reduction of particle generation during the

Every revolution is, well, revolutionary. Processes and people massively change in a way that rocks the
ISPE’s Pharma 4.0 initiative points to everything the pharmaceutical industry could be if it embraced the

For years now, robots and automation have been making their way into pharmaceutical production. Lately, though,
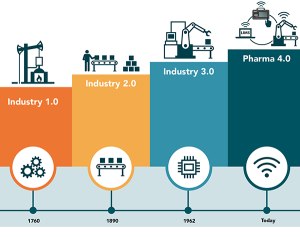
A lot has changed since the 1700s in the whole world, but especially in the pharmaceutical

Your Contamination Control Strategy (CCS) is the foundation of your cleanroom. This is where you plan

The European Union (EU) Good Manufacturing Practice (GMP) for Medicinal Products for Human and Veterinary Use

We talk a lot about data integrity, but what happens, in the real world, when US
It’s not a secret: PPE is important in every healthcare setting. But in the intensity of
Do you remember the last time you went to a show where there was a “splash
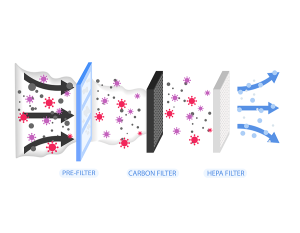
No one goes into medicine to cause harm. Doctors, nurses, techs, administrators, PAs, CNAs, and all
Did you know that approximately 1 out of every 31 hospital patients in the U.S. are
USP <800> provides guidelines that “describes requirements including responsibilities of personnel handling hazardous drugs; facility and

Our furry family members sometimes go down for the count and need some medication to help

Whether you are a new compounding pharmacist or have been around for a while, you know

You probably don’t think of pharmacies as cleanrooms or controlled environments, but that’s because you are

In 2020, a vote found EN ISO 14698 no longer suitable for use in Europe, and,

After 17 years of no revisions, a formal vote in 2019 replaced EN ISO 14698 with

You will hopefully not be shocked to learn that data integrity is an integral part of

At the start of 2022, we outlined some trends we expected to see grow in popularity

Have you ever heard the old wives’ tale that if you water your garden at night,

During the 2020 fire season, more than 106 large wildfires occurred in the states of Oregon

EN 17141 Cleanrooms and controlled environments – Biocontamination control is a relatively new European standard, introduced

An airborne particle counter only needs to know two things to begin sampling: where it is

What is a foundational piece of your cleanroom contamination control strategy? Your gowning protocol. Humans, by

An active air sampler is one of the tools you can use in your cleanroom to

This year, Lighthouse Worldwide Solutions is celebrating its 40th anniversary. Over the last 40 years, we

In 2017, GMP released a draft update to Annex 1, which received a lot of feedback.
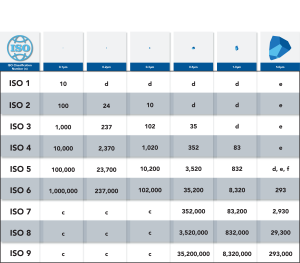
A cleanroom classification basically tells you how clean a cleanroom is. While we typically consider cleanrooms